Bacteria-killing protein could lead to a new treatment for diabetes
Posted: 24 February 2016 | Victoria White | No comments yet
Researchers have discovered that the protein cathelicidin antimicrobial peptide helps the pancreas regenerate and produce insulin…
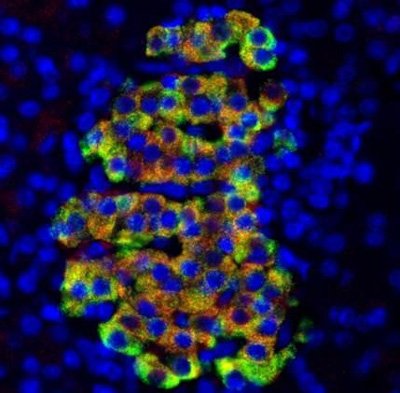

Insulin appears green while Cathelicidin antimicrobial peptide (CAMP) appears red and the DNA of all cells appears blue. CREDIT: Dr Fraser Scott, The Ottawa Hospital
For some time, scientists have puzzled over the presence of a bacteria-killing protein in an area of the body that is not normally exposed to bacteria – the pancreas.
But now, researchers at The Ottawa Hospital and the University of Ottawa have discovered that the protein in question was doing something entirely unexpected – it was actually helping the pancreas regenerate and produce insulin. This ground-breaking discovery could lead to new treatments for this devastating disease.
Dr Fraser Scott and his team study type 1 diabetes, which occurs when the immune system mistakenly attacks and destroys insulin-producing cells in the pancreas. Nobody knows exactly what causes this, but genetics, diet, bacteria and viruses are thought to play a role.
Dr Scott’s previous research suggested that a bacteria-killing protein called cathelicidin antimicrobial peptide (CAMP) might also play a role in type 1 diabetes, but it was not clear how.
“We were looking for this bacteria-killing protein in various parts of the body, and as expected, we found high levels in the gut tissues that are exposed to bacteria,” explained Dr Scott, a senior scientist at The Ottawa Hospital and professor at the University of Ottawa. “However, we also found it in the pancreas, which was a complete shock because the pancreas isn’t typically exposed to bacteria.”
CAMP gene expression is lower in diabetes-prone rats
Dr Scott and his team did further experiments to try to determine what CAMP was doing in the pancreas and how it might be linked to diabetes. They found that CAMP is produced by the same pancreatic cells that produce insulin in rats, mice and humans. They also treated pancreatic cells in the laboratory with CAMP, and found that it doubled insulin secretion. When they injected diabetes-prone rats with CAMP, they also found signs of increased regeneration in the pancreas and a shift towards more beneficial bacteria in the gut. They also found that CAMP gene expression was lower in diabetes-prone rats than in normal rats, highlighting a potential opportunity for disease treatment.
“Our study uncovers an intriguing new role for this protein in pancreas function and regeneration, with possible links to diabetes-associated gut bacteria,” said Dr Scott. “We certainly don’t have all the answers yet, but our findings raise the exciting possibility of novel treatments for both type 1 and type 2 diabetes.”
Related conditions
Diabetes



