FDA grants orphan drug designation to BLU-554
30 September 2015 | By Victoria White
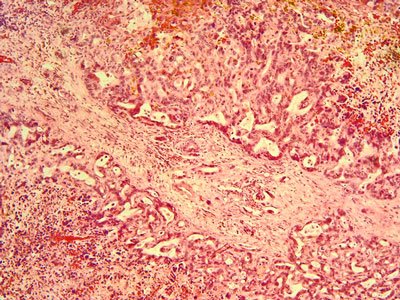
BLU-554, an exquisitely selective inhibitor of fibroblast growth factor receptor 4 (FGFR4), is currently being evaluated in a Phase 1 clinical trial in patients with advanced hepatocellular carcinoma...