Discovery of molecular protection linked to Kennedy’s disease
Posted: 10 June 2016 | Victoria White, Digital Content Producer | 7 comments
The discovery brings in-depth molecular insights that can bring researchers closer to finding a therapeutic target for Kennedy’s disease…
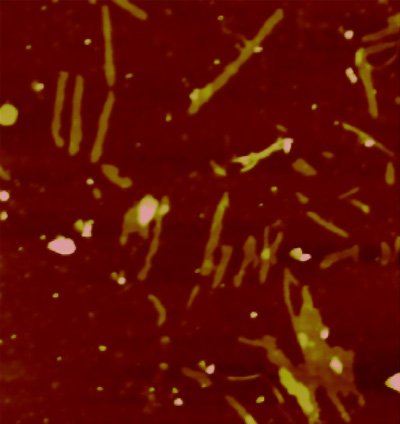

Protein Androgen Receptor forms aggregates that damage muscle and motor neuron cells in Kennedy’s Disease. CREDIT: B. Eftekharzadeh, IRB Barcelona
Scientists from IRB Barcelona have described a molecular system of protection that involves the androgen receptor protein, a molecule that is mutated in patients with Kennedy’s disease and which cause progressive muscle wastage.
The finding brings in-depth molecular insights that can lead to new studies and bring researchers closer to finding a therapeutic target for Kennedy’s disease.
In Kennedy’s disease, the muscle cells and motor neurons are damaged as a result of the accumulation of androgen receptor fibres—a process that causes them to die. “Many aspects of diseases involving aggregates, such as Alzheimer’s and Parkinson’s, are unknown. In this regard, Kennedy’s disease is in a worse position because it is a rare condition,” explains Xavier Salvatella, head of the Molecular Biophysics Lab at IRB Barcelona.
The onset of this genetically inherited disease occurs in late adulthood, affecting one in every 40,000 men and causing progressive deterioration of all muscles. Although not fatal, the condition is debilitating, and 20% of those affected eventually need a wheel chair.
Salvatella studies the sequence of molecular events that causes the aggregation of androgen receptors. He and his team seek to determine the regions of the protein sequence responsible for aggregation in order to identify valid targets to direct therapies and to prevent this process.
The mutation carried by those affected by Kennedy’s disease causes a repeated and excessively long chain of a specific amino acid, namely glutamine, which impairs the activity of the androgen receptor. This protein, which activates the hormone testosterone, is responsible for triggering the genetic programme that favours the differential characteristics of men.
During adolescence, and depending on the extent to which they are affected, boys with the mutation do not fully develop the male phenotype, but this is often not diagnosed. In a second stage, in adulthood, muscle degeneration begins.
Leucine residues
Thanks to access to one of the main Nuclear Magnetic Resonance facilities in Europe, located at the University of Florence, for the first time the scientists have studied the protein in a test tube. They have observed that right next to the glutamine chain there is a region comprised by four leucine residues that allay the effects of the mutation.
The leucine molecules favour the folding of the polyglutamine chain into a helix, a structure that prevents the chains from adhering to one another. However, the impact of the leucine molecule on the glutamine region is limited, and if there are many glutamine amino acids, the chains do not fold. Instead, they stretch out like rods, stick to each other, and end up forming a fibrous wall.
“We have seen that four leucine molecules delay this process. What would happen in the presence of six?” asks Salvatella. “Conceptually speaking, one clever way of delaying the aggregation could be to use drugs to strengthen the effect of the leucine residues that have so much influence on the mutation site that causes the protein to aggregate.”
Related topics
Amino acids
Related conditions
Kennedy's disease




This seems to be awesome news!!! God willing and good researchers maybe soon there will be a treatment. I know everyone is working hard to find a treatment!!! Keep up the good work!!
I am 71, diagnosed at Mayo Clinic in 2014 after being misdiagnosed as LGMD in 2007. I had no physical symptoms until I was 64. That was minimal, only flag was high cpk of 1900. My mother who is 90 tested positive as carrier. She had 8 brothers none of which had any noticeable symptoms nor did her father. I had two things that I believe caused my Kennedy to manifest, I fasted 40 days liquid only in1996. I believe lose of muscle wasn’t regained because of Kennedy. I am convinced that large doses of Leviquin antibiotics accelerated problem. In 2005 had 30 day dose for prostate infection, 2006 one week for infection of muscle biopsy, but the killer for sure was 40 days of Leviquin by iv for knee infection following knee surgery, then 60 days by mouth. Drastic change in my strength as a result. Now I walk with walker or cane and that is limited
EXTRAORDINARY NEWS……OUR APPRECIATION FOR ALL SCIENTIST
WORKING IN THE VARIOUS EFFORTS TO GET A CURE FOR OUR DISEASE.
BIG THANKS FROM MY HEART..
MARIO
BIG THANKS FOR ALL THE SCIENTIS WORKING TO GET A CURE FOR OUR DISEASE…..FROM MY HEART….MARIO
Excellent news. Thanks to all the doctors and scientists for their efforts in finding a cure to this disease.
If there are any drug trials or other studies where those of us with Kennedy’s Disease are needed, I’d be very interested in participating. I am 62 and was diagnosed via DNA blood test about 7 months ago.
God bless all those who fight this disease, I diagnosed 6 years ago and there two other brothers also with this, My mother had MS and its was hard to see her waste away from normal to almost bed ridden. If there is any way to get on new trail for any new treatment, for Kennedy disease, please put my name in . I go to U of M here in Mi and would like to see great progress for this disease, I was blessed with two daughters and worry about their future and offspring. If possible, please let me know if there is any new way to get on the new treatment. God Bless to all of those working to find a break through. James
Thank God for the progress already made towards finding a cure for Kennedy Disease.I was diagnosed in 1986? Now movement and balance are impaired.I need support on both sides to walk. Is there any further encouraging development to keep us hoping??